Giving back the power of speech
From a bold vision at the Wyss Center to a company on the path to First-in-Human — this is the story of ABILITY Neurotech.
The vision is born
Professor John Donoghue, widely regarded as the father of brain-computer interfaces and founding director of the Wyss Center for Bio and Neuroengineering in Geneva, conceives the idea of a fully implantable speech neuroprosthesis.
His vision: a device that could decode neural signals directly into natural speech, giving a voice back to patients silenced by neurological conditions.

Proving the impossible
In a landmark clinical case study, Wyss Center scientists (now ABILITY) worked with a fully paralysed person living with amyotrophic lateral sclerosis (ALS) — a progressive neurodegenerative disease in which patients lose the ability to move and speak.
Using implanted electrodes, the patient was able to communicate by modulating his neural activity to produce yes/no responses for more than two years in a home set-up, which he used to select letters and spell out complete sentences.
These efforts demonstrated the remarkable potential of chronic brain-computer interfaces to restore lost function in complete locked-in state. This work was published in Nature Communications.
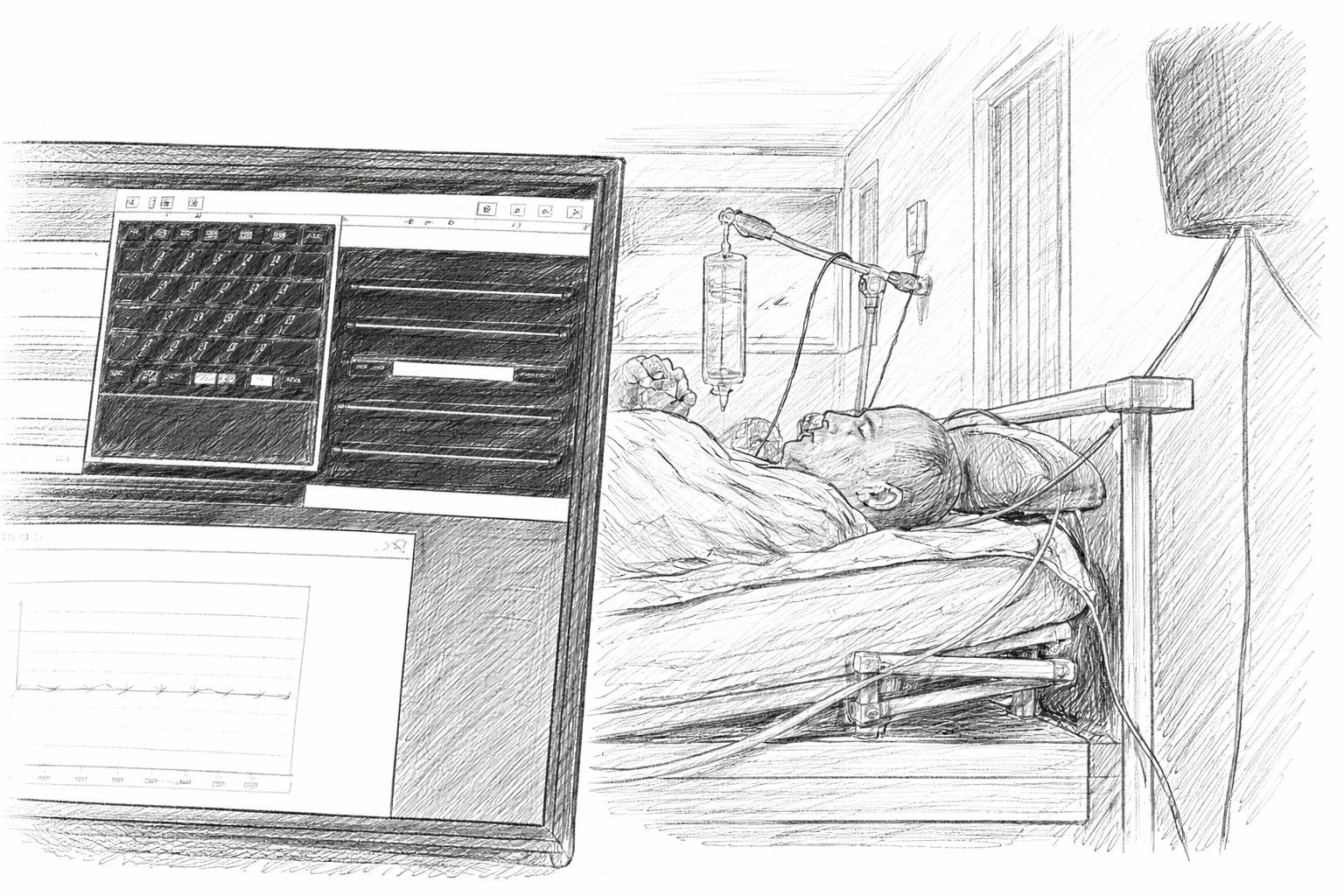
Pioneering research at the Wyss Center
Under the umbrella of the Wyss Center, the project grows into a world-class research programme. A multidisciplinary team of neuroscientists, engineers, and clinicians develops the core technology — from implantable electronics and optical data links to advanced neural decoding algorithms.
The programme achieves major scientific milestones, including several pre-clinical trials in sheep and monkey models that validate the safety and performance of the device. These results lay the scientific and clinical foundation for what comes next.
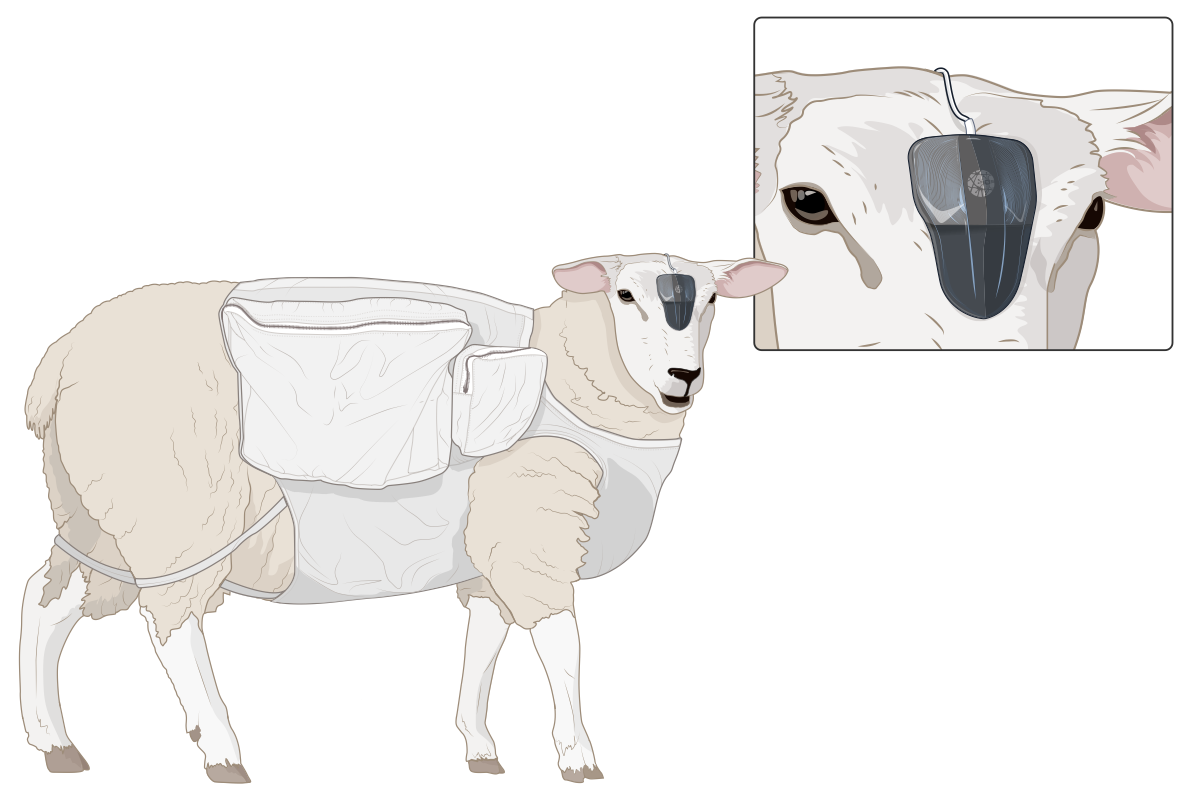
ABILITY Neurotech is born
After years of breakthrough research, the project spins out from the Wyss Center as ABILITY Neurotech — an independent company with a clear commercial mission: to bring a fully implantable brain-computer interface to market that restores communication for people who have lost the ability to speak.
Based at Campus Biotech in Geneva, the founding team brings together deep expertise in neuroscience, medical device engineering, machine learning, quality, and regulatory affairs.

First-in-Human technical file submitted
After completing a rigorous verification campaign — including environmental stress tests, impact testing, electrical verification, and device soak trials — ABILITY submits its Investigational Medical Device Dossier (IMDD) technical file, marking a decisive step toward clinical trials.
In parallel, ABILITY submits a Breakthrough Device Designation (BDD) request to the U.S. FDA, reflecting the novel nature of the technology and its potential to address a critical unmet clinical need.

Next stop: First-in-Human
ABILITY is preparing for its First-in-Human (FIH) clinical trial, to be conducted within the INTRECOM project — a European collaborative initiative to advance implantable speech BCIs into clinical practice.
The trial will be carried out in collaboration with TU Graz (Austria) and UMC Utrecht (Netherlands), who will serve as clinical sponsors. This milestone will mark the first time ABILITY's fully implantable speech neuroprosthesis is tested in a human patient.

Our Mission
To develop and deliver a fully implantable brain-computer interface that restores natural speech for patients who have lost the ability to communicate, enabling them to reconnect with their loved ones and the world.
Our Vision
A world where neurological conditions no longer silence the human voice. We envision speech neuroprostheses as a standard of care — safe, reliable, and accessible to every patient who needs one.